
APACERAM / APACERAM-AX
Biocompatible ceramic bone graft substitute APACERAM was created by our own unique technology. Approved products, models and indications may differ from country to country. Please contact us for detail.

- category:Bone Graft Substitutes
- feature:Non-absorbable
APACERAM / APACERAM-AX
Hydroxyapatite [Ca10(PO4)6(OH)2] has excellent biocompatibility and biological safety, and is available in a wide variety of APACERAM and ultra-high porosity APACERAM-AX with a porosity of 85% with a triple pore structure.
APACERAM
Biocompatible ceramic bone graft substitute APACERAM was created by our own unique technology.
Approved products, models and indications may differ from country to country.
Please contact us for detail.
APACERAM-AX
APACERAM-AX is a bone replacement material with a characteristic microstructure originally developed by our company, and means “new axial attachment (AXIS), highest (MAX) porosity, and highest (MAX) osteogenic capacity” in the APACERAM product group.
Features of APACERAM
Biocompatibility
APACERAM consists of synthetic hydroxyapatite 〔Ca10(PO4)6(OH)2〕that is an inorganic component of human bone. It has excellent biocompatibility and biological safety.
Osteoconductivity
APACERAM has macro pores and micro pores in the structure. Macro pores are effective for new bone formation, while micro pores provide interconnectivity of the pores.
Wide variation
Wide varieties of the models are available with various mechanical strength and porosity depending on the application sites and intended use.
Feature of APACERAM porous materials
SEM image of porous surface (porosity 50%): Continuous porosity

Undecalcified sample of APACERAM that has been placed in the femur of adult canine (3 months after implantation).New bone was observed intruding into pores of APACERAM.
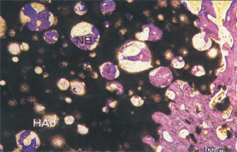
Feature of APACERAM (dense material)
SEM image of the dense material (surface)
High density sintered body consisting of closely packed particles of 0.3~0.5μm.

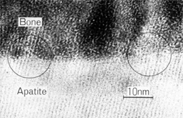
TEM image of the interface between APACERAM (dense material) and the bone【Photo】 Dr. Makoto Ogiso (Tokyo Medical Dental University) Crystalline particles are continuously emerging in the same direction at the interface between bone and APACERAM. It confirms complete chemical bonding between bone and APACERAM. (TEM image:2 months after implantation)
Porosity and strength
| Porosity (%) | 3-point flexural strength MPacompressive | strength MPa |
| 0 | 220 | 730 |
| 5 | 90 | 540 |
| 15 | 50 | 240 |
| 30 | 27 | 175 |
| 40 | 16 | 65 |
| 50 | 10 | 30 |
| 55 | 9 | 20 |
| 60 | 7 | 15 |
Product Line-up (APACERAM)
A variety of porosity and shapes are available depending on the intended use and implantation site, including versatile blocks, granules and various spacers.
*Please refer to the catalog for details. (Please refer to the product catalog page)

Features of APACERAM-AX
Triple pore structure
This is an ultra-high-porosity hydroxyapatite artificial bone with 85% porosity and a triple-pore structure:① Spherical macro pores, ② Considerable interconnecting pores, ③ micro pores among the spherical particles

Assimilation with bone tissue*
It bonds with the surrounding bone as new bone grows inside its pores.
Implanted in the defect Ø4mm × 6mmL in the iliac of the beagle (radiography).

In 26th weeks after implantation, boundaries around the material became vague showing progress of new bone formation.
* To become almost same situation as bone tissue in terms of structures and functions.
Early osteogenic ability
Interconnecting pore structure and porous inner wall structure of the pores enable early bone formation.
a) Implanted in the defect Ø4mm × 6mmL in the iliac of the beagle (Toluidine blue staining).
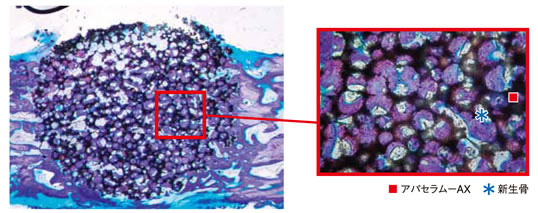
In 4 weeks after implantation, new bone formation was observed all over the implanted site with active new bone in the pores.
b) Implantation of APACERAM-AX in the defect Ø4mm × 12mmL in the iliac of the beagle. (Hematoxylin Eosin staining)
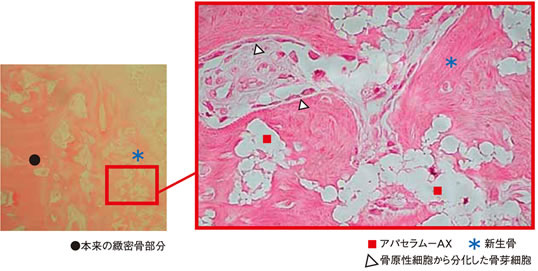
In 4 weeks after implantation, new bone formation was observed in the pores and osteoblasts were found on the surface of the newly formed bone.
Product Line-up (APACERAM-AX)

| Product number | name | classification, name, abbreviated | packaging unit/box |
| GA-2 | Granules 1.0-2.0mm | Artificial Bone, AB-01 | 2g |
| GA-3 | Filler 3.0±1.0mm | Artificial Bone AB-01 | 2g |
| A-1 | block 10×10×10mm | artificial bone, AB-02 | 1ml×1 Piece |
| A-2 | block 10×10×20mm | artificial bone, AB-02 | 2ml×1 Piece |
| A-3 | PLF Stick 4×4×40mm | Artificial Bone / AB-13 | 1 |




