


“ReFit” is a bone graft substitutes made of hydroxyapatite and collagen composite.
Approved products, models and indications may differ from country to country.
Please contact us for detail.
※Compared with the similar products when filled in bone defects caused by bone disease like bone tumor, trauma or after harvesting autograft.

| Components | Inorganic component:Low crystalline Hydroxyapatite Organic component: Type I collagen (derived from porcine dermis) |
|---|---|
| Composition (weight ratio) |
Hydroxyapatite (HAp):ca 80wt% Collagen (Col):ca 20wt% |
| Porosity | 95% |
| Pore diameter | 100−500μm |
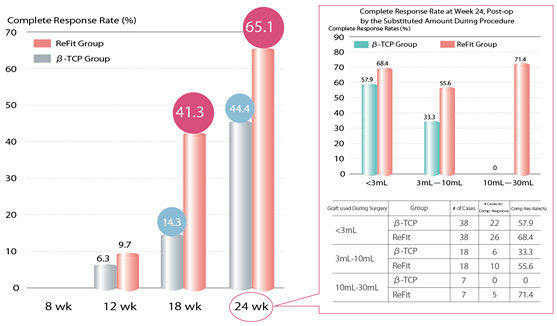
Clinical trial of ReFit was conducted in accordance to “Guidelines for clinical test for medical devices (Medical device GCP)”
Comparative study was performed using a mormal β-tricalcium phosphate as a control device for absorbable bone graft.
■Period of observation:24 weeks post operation
■Facilities for clinical trial: seven facilities
■Number of cases: ReFit 63 cases, β-TCP:63 cases
Evaluation of efficacy
Radiographic study revealed that ReFit group was superior to β-TCP group regarding bone formation in 18 weeks and thereafter. In 24 weeks, remarkable efficiency rate of ReFit group by filling volume was 50% or higher for every filling volume irrelevant to the volume (71.4% for filling volume 10-30mL). On the other hand for β-TCP group, remarkable efficiency rate decreased with increasing the filing volume (0% for filling volume 10-30mL)

| Product code | PB-101010 | PB-103020 |
|---|---|---|
| Size | 10×10×10mm | 10x30x20mm |
| Qty/package | 1 pc(1.0mL) | 1pc(6.0mL) |
【Contraindications and prohibitions】
・DO NOT reuse
・Allergic subjects with medical history of anaphylaxis, severe allergic subjects, or patients with allergy against drugs containing porcine collagen.